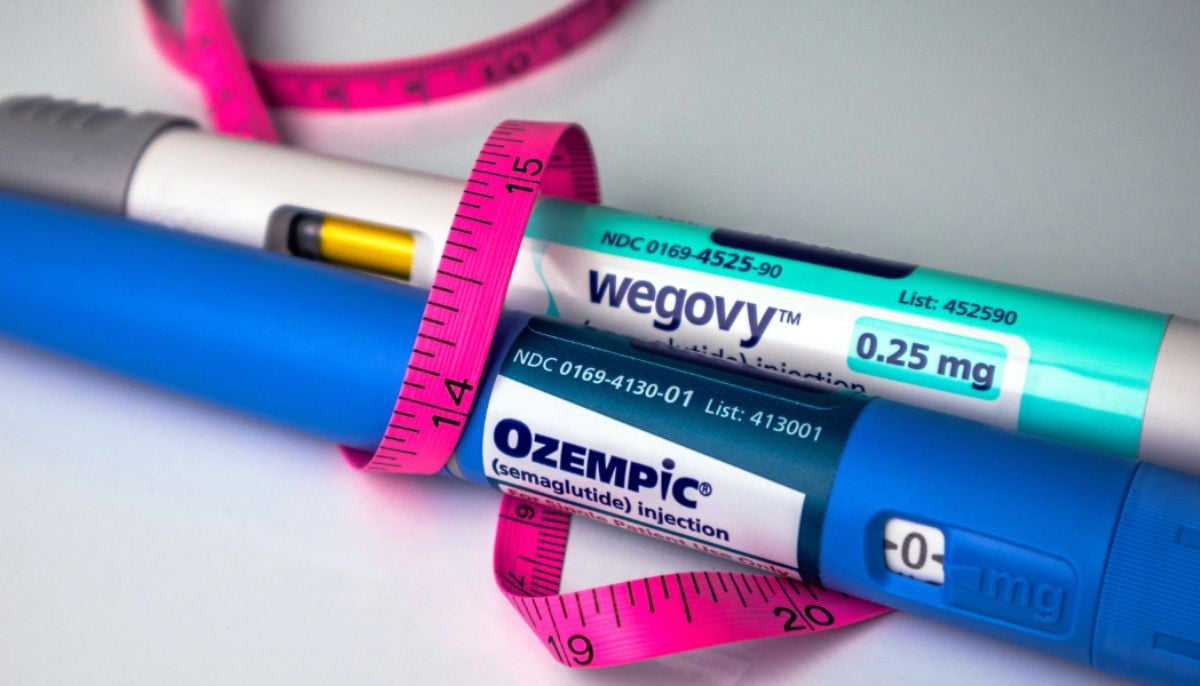
Some Chinese companies are now racing to make generic versions of Novo Nordisk’s Wegovy, also supplied ingredients for more than a billion improvised doses of weight loss medicines that have been sold online in the US for the past two years, according to three sources and a reutures evaluation of shipping and public registers.
Cheap copies of Wegovy and Eli Lilly’s Zepbound are on the retreat in the US because supervisors limit their sale, and the shipments of Chinese suppliers of the rough ingredients delayed the explosive growth of the medicines.
Robert Califf, who had two stints that led the American Food and Drug Administration, said that never had a new medicine before that became so hugely popular that the manufacturer just couldn’t keep up.
The shortage opened the door for putting together pharmacies, turbocommressor by Telehalth companies that flourished during the COVID Pandemie, to deliver cheap copies to a huge market that chases the promised weight loss.
The pivot to generic medicines approved by the FDA as patents in different countries expire a year of increasing demand for brand drugs, which has been shown to help people to gain up to 20% of their weight.
At least eight Chinese companies, including listed Jiangsu Sinopep-Allsino Biofarmaceutical and Hybio-Pharmaceutical helped the US flood with rough semaglutide and Tirzepatide, the most important ingredients in Wegovy and Zepbound, told VRONTERS. A Reuters analysis of the American FDA data data supports that.
Hybio and Sinopep are working on launching their own generic semaglutids, according to one of the sources with knowledge of the efforts and earlier Reuters reporting. The source also said that Nanjing Hanxin Pharmaceutical and Fujian Genohope Biotech, two of the companies that had supplied compounders, also launch.
None of the companies responded to requests for comments.
American legislation now limits the composite pharmacies to produce personalized doses for patients they need, or formulations that are not offered by the brand medicines.
The source with knowledge of the Hybio and Sinopep efforts said that for a Chinese manufacturer, delivering ingredients for composite weight loss medicines had been the largest company. The manufacturer is now focusing on markets where NOVO’s most important semaglutide patent, such as Canada and Brazil, will expire next year to sell the ingredient to generic medicine makers, he said.
He said that he was aware of at least five other Chinese companies that are known to deliver compound pharmacies that re -concentrated in the same way on supplying semaglutide for generic medicines.
It is unlikely that the switch will produce similar explosive growth. The production of Semaglutide to his last, injectable form is complex, said a lawyer for a generic drugstore who asked for anonymity because he was not authorized to discuss the subject.
Companies that want to sell generics can also conclude deals with branded companies that postpone their market input.
‘One -off issue of a decade’
Increasing the much -needed drugs for weight loss, while the brand medicines were scarce, offered an unprecedented opportunity.
In 2024 alone, the eight Chinese companies sent enough raw material to the US to produce more than 1 billion starter doses of the Blockbuster Medicines, according to FDA -shipping data.
The estimate of Novo is even higher. It said that Chinese companies shift enough semaglutide to the US in just over six months to make 1.5 billion starter doses Wegovy, according to a letter submitted to the US Department of Trade and publicly posted.
In the first quarter of this year, with the range of business offering no longer a problem and the FDA that insists on an end to massage demonstration, Hybio, Sinopep and others still brought in significant shipments of ingredients to copy both drugs.
By the second quarter, shipments had fallen 90% compared to a year earlier for Semaglutide, also the main ingredient in the Diabetes Treatment of NOVO, and with 34% for tirzepatide used to make Zepbound and Mounjaro copies.
The economy for Chinese companies that sold semaglutide to compounders was attractive. A month of Semaglutide -powder’s range only costs 7 cents to produce, according to a report from 2024 in Jama. Chinese ingredient makers can sell this for a maximum of seven times that amount to manufacturers who want to make copies, based on sales figures provided by the source.
American composite pharmacies sold the injectable medicines for an average of only $ 230 a month, more than half of brand prices.
The costs for NOVO have been high. It missed quarterly sales goals and shares of the Danish drug maker have been halved this year, which contributes to the Treater of the CEO.
Data shows that Sinopep, Hybio and another company have started sending to the US. More Liraglutide, the most important ingredient of an older Novo medication that is sold under the brand names Victoza and Saxenda that leads to more modest weight loss.
Generic versions of Liraglutide, available in the US since 2024, are now characterized by Online TeleHealth sites that once have pushed semaglutide.
A second source, a wholesaler that sells obesity drug ingredients to putting together pharmacies, but that was not authorized to speak publicly, said he had seen an increase in the sale of liraglutide.
Marta Wosinska, a senior fellow at the Brookings Institution that followed the rise of this industry, said that Chinese shipments made Semaglutide had announced the deficits in 2022 not long after the FDA had announced.
Califf said he expected companies to test the law that limits the sale, and that the FDA will set the tone for enforcement with industrial guidelines. The FDA said that it is committed to using all available tools to supervise the safety and quality of products regulated by the FDA.
Still, Califf said, compiling weight loss drugs on a huge scale is probably a “one -off problem”.